Make an Igloo
Take the quiz and check your answers!
 Click here to take the quiz!
Click here to take the quiz!All About Snow

 shape. This hexagon shape forms because, when liquid water turns into solid ice, the water molecules arrange themselves in a hexagon pattern.
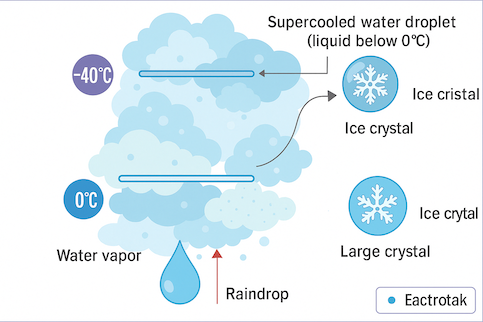
Snow usually forms in cold and wet clouds. Inside these clouds, which are colder than 0°C, there are tiny water droplets that haven’t frozen yet (called supercooled droplets), small pieces of ice, and water vapor. Water vapor sticks to the ice crystals, and the snowflake starts to grow. When ice crystals bump into other ice or droplets, they become bigger and heavier and then fall to the ground.
The shape of a snowflake depends on the ②
shape. This hexagon shape forms because, when liquid water turns into solid ice, the water molecules arrange themselves in a hexagon pattern.
Snow usually forms in cold and wet clouds. Inside these clouds, which are colder than 0°C, there are tiny water droplets that haven’t frozen yet (called supercooled droplets), small pieces of ice, and water vapor. Water vapor sticks to the ice crystals, and the snowflake starts to grow. When ice crystals bump into other ice or droplets, they become bigger and heavier and then fall to the ground.
The shape of a snowflake depends on the ②  and ③
and ③  in the cloud. Usually, when the temperature is between -10°C and -20°C and the humidity is high, snowflakes form beautiful, complex shapes with many branches.
.So, are artificial snowflakes made at ski resorts as beautiful as natural ones? Artificial snow usually has simpler crystal shapes than natural snow. To make artificial snow with a ④
in the cloud. Usually, when the temperature is between -10°C and -20°C and the humidity is high, snowflakes form beautiful, complex shapes with many branches.
.So, are artificial snowflakes made at ski resorts as beautiful as natural ones? Artificial snow usually has simpler crystal shapes than natural snow. To make artificial snow with a ④  , the air temperature must be between -2°C and -3°C, and the humidity should be less than 60%. The snowmaker sprays fine water droplets into the air at high pressure, and they freeze right away. Because artificial snow does not form through repeated collisions of ice crystals like natural snow, its crystals are simpler in shape.
, the air temperature must be between -2°C and -3°C, and the humidity should be less than 60%. The snowmaker sprays fine water droplets into the air at high pressure, and they freeze right away. Because artificial snow does not form through repeated collisions of ice crystals like natural snow, its crystals are simpler in shape.

Making artificial snow with a snowmaker

Artificial snow crystal
 Click here to take the quiz!
Click here to take the quiz!The Three States of Water and How They Change
Observing the States of Water
Water exists in three states: solid (ice), liquid (water), and gas (water vapor). Observe the ice, water, and vapor and record their characteristics.
Classifying Water Substances by Their State
Let’s sort different substances made of water into their correct state.

Snow

Icicles

Glaciers

Frost

Fog on Glasses

Mist

Clouds

Dew

Rain

Bubbles in Boiling Water
Observing Water’s Change of State
When you freeze water in a plastic bottle, its shape changes, as shown in the picture. Explain why a water jar left outside in the cold winter breaks, based on the change in the state of water.
 Click here to take the quiz!
Click here to take the quiz!Why Is It Warm Inside an Igloo?
Activity 1
Please follow the instructions and assemble the educational kit in the order specified.
Activity 2: Observe and classify the state of water around the igloo
Look at the picture below and find all the things made of water around the cold polar igloo. Then, write them under the categories: solid, liquid, or gas.
Activity 3: Infer Like a Scientist
We can use changes in the state of water to make the temperature go up or down. When Water changes into gas (water vapor), it absorbs heat, which makes the air cooler. In contrast, when water changes into solid (ice)), it releases heat, which makes the air warmer. Look at the two situations below and write which state change of water is happening.








 (B) In Korea, people spray water in the yard on hot summer days to cool down the temperature.
(B) In Korea, people spray water in the yard on hot summer days to cool down the temperature.